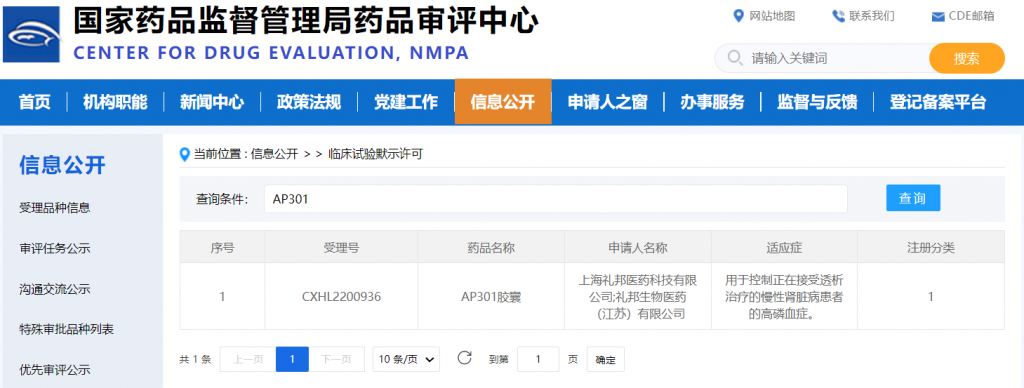
March 17, 2023, Shanghai, China—Alebund Pharmaceuticals (“Alebund” or the “Company”) announced that China’s National Medical Products Administration (NMPA) has granted approval for the company’s IND application for a phase 3 study of AP301 to treat hyperphosphatemia in dialysis patients.
AP301, a new-generation iron-based phosphate binder, is a class Ⅰ drug developed by Alebund for the treatment of hyperphosphatemia in patients with renal failure. Through binding to phosphate in the gastrointestinal (GI) tract and reducing GI phosphate absorption, AP301 decreases serum phosphorus levels and improves the long-term outcome of patients with renal failure. This is a phase 3, randomized, open-label, multicenter study to evaluate the efficacy and safety of AP301 on serum phosphorus control in chronic kidney disease patients receiving maintenance dialysis with hyperphosphatemia. In the previously completed phase 1 and phase 2 studies, AP301 showed an efficacious effect on lowering serum phosphorus in the same population and was safe and well tolerated.
“More than eight hundred thousand patients are receiving dialysis in China, and the number continues to grow rapidly. Less than half of them whose serum phosphorus levels are under control, there is a pressing unmet medical need.” said Dr. Jin Tian, CMO of Alebund, “We hope that the phase 3 study will progress smoothly, and we can soon provide a safe and efficacious treatment option to the patients.”
“With the company’s most clinically advanced products entering the late-stage clinical development, Alebund has been actively preparing on the manufacturing end. Our research and production facility in Yangzhou, China, would lay a strong milestone on our path to launch and commercialize Alebund’s pipelines in China and overseas.” Gavin Xia, CEO of Alebund said, “The recent advancements in our pipelines and the construction of the Yangzhou facility demonstrate Alebund’s dedication and commitment to the renal disease area.”

About Hyperphosphatemia
Hyperphosphatemia is one of the most common complications in patients with chronic kidney disease. Chronically high blood phosphorus levels can cause multiple complications e.g., secondary hyperparathyroidism, renal osteodystrophy, and vascular calcification. It is an independent risk factor of cardiovascular risk and all-cause mortalities. Controlling serum phosphorus can effectively improve the patient’s outcome with chronic kidney disease.
About Alebund Pharmaceuticals
Alebund is a startup jointly incubated by a group of industry leaders in the field of nephrology in Shanghai in 2018. As a clinical-stage biopharmaceutical company, Alebund focuses on the discovery and development of novel therapies primarily for kidney diseases and their complications, as well as other chronic conditions. Alebund has built a diversified and balanced pipeline of drug candidates targeting a range of renal diseases, including chronic kidney disease (CKD)/dialysis complications, IgA nephropathy, diabetic kidney disease, and autosomal dominant polycystic kidney disease (ADPKD). Alehund’s pipeline comprises both small-molecule and biological assets.
礼邦医药治疗透析患者高磷血症的新一代磷结合剂 AP301 获准在中国开展 Ⅲ 期临床试验
2023 年 3 月 17 日,中国上海——礼邦医药(Alebund Pharmaceuticals)宣布,近日已获得中国国家药品监督管理局(NMPA)核准签发关于 AP301 的《药物临床试验批准通知书》,同意本品开展 Ⅲ 期临床试验。
AP301 是一款新型含铁磷酸盐结合剂,为礼邦医药自主研制开发的一类新药,拟用于治疗慢性肾功能衰竭患者的高磷血症。AP301 在肠道内高效结合食物中摄入的磷酸盐促进磷从肠道排出而减少磷的吸收,从而降低患者血磷,改善慢性肾功能衰竭患者的长期临床结局。此次获准开展的临床试验为一项在慢性肾脏病接受维持性透析的高磷血症患者中评价 AP301 控制血清磷的有效性和安全性的随机、开放、多中心 Ⅲ 期研究。此前,该药物已成功完成 Ⅰ 期和 Ⅱ 期临床试验,显示了其在透析患者中确切的降磷效果,且拥有良好的安全性和耐受性。
“我国目前接受透析患者已达 80 余万,且仍在快速增长;而这些患者血磷达标率不足 50%,有巨大未满足的临床需求,”礼邦医药首席医学官田劲医生表示,“我们期待这项临床研究顺利进行,早日为患者提供安全有效治疗高磷血症的新的药物。”
“随着公司管线产品进入临床后期,礼邦已经开始积极筹备产业化落地。中国扬州的生产基地将为 AP301 等产品在国内外上市及销售提供坚实的基础。”礼邦医药首席执行官夏国尧博士说道,“公司近期多个临床管线的阶段性推动和产业化基地的建设,也充分证明了礼邦团队对肾脏病领域的长期坚守和充分信心。”
关于高磷血症
高磷血症是慢性肾脏病患者得重要并发症之一。血磷水平长期过高可导致甲状旁腺功能亢进、肾性骨病、血管钙化等多种并发症,是增加患者心血管事件和全因死亡的独立危险因素。控制血磷水平达标可有效改善慢性肾脏病患者的预后。
关于礼邦医药
2018 年初,礼邦医药由最顶尖的肾脏病领域行业领导者孵化于中国上海,目前是一家处于临床阶段的生物制药公司,主要致力于肾脏病以及其他相关慢性疾病的创新药物发现和开发,为慢性肾脏病及相关疾病患者提供更佳临床治疗方案。礼邦医药已经建立起了丰富且均衡的肾脏病新药产品管线,包括针对慢性肾病(CKD)/透析并发症、IgA 肾病、糖尿病肾病、常染色体显性多囊肾病(ADPKD)等产品,公司在研产品包括小分子药物和生物制剂。